 The pink arrow indicates the direction of the laser beam. C: Picture of the experimental setting sp, speaker. 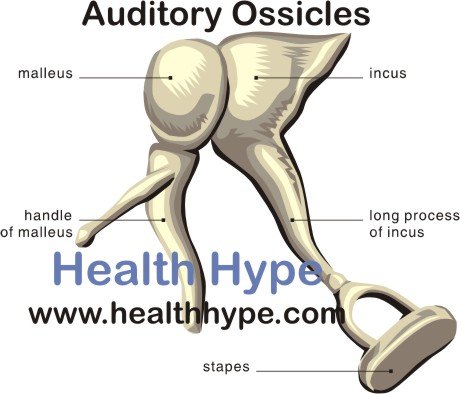
* P < 0.05 and ** P < 0.001 versus wild-type controls. Rankl −/− mice ( A, open squares, aged 6 to 8 weeks, n = 6) and Fos −/− mice ( B, open squares, aged 6 to 8 weeks, n = 6) were compared with wild-type controls (closed squares, aged 6 to 8 weeks, n = 6 for each). A and B: Evaluation of the hearing threshold based on auditory brainstem response. Biomicroscopyįigure 1 Hearing loss and impaired vibration of the malleus in osteopetrotic mice. Because the tympanic cavity is continuous anteriorly with the Eustachian tube (auditory tube) and posteriorly with small cavities filled with air (mastoid air cells), we defined anterior and posterior limits of the tympanic cavity arbitrarily based on the anterior end of the malleus and the posterior end of the stapes, respectively, so that the entire array of ossicles is encased. Superiorly, the tegmen tympani forms the roof and, inferiorly, the bone over the jugular vein forms the floor of the middle ear. The tympanic membrane forms the lateral wall, whereas the promontory (a part of the otic capsule) forms the medial wall. For volumetric analysis of wild-type and osteopetrotic mice, the limits of the tympanic cavity were defined as follows. The three-dimensional (3D) data were analyzed using computer software (Tri/3D-BON software Ratoc System Engineering, Tokyo). Images were 1024 × 1024 × 1024 pixels at a voxel size of 4.93 μm. In several experiments, a μCT scanner (model TDM1000 Yamato Scientific, Tokyo) was operated at 80 kV and 35 μ A using 16 integrations and 800 projections. Reconstructed images were 481 × 481 × 481 pixels at a voxel size of 10 μm. The CT images were reconstructed from 512 projections using computer software (i-view-R software J. A μCT scanner (R_mCT Rigaku Corporation, Tokyo, Japan) was operated at 90 kV and 150 μA over 120 seconds. The skulls of Rankl −/−, Fos −/−, Src −/−, and control mice were imaged using microcomputed tomographic (μCT) scanners. 
These data suggest that osteoclastic bone resorption is required for thinning of auditory ossicles and enlargement of the tympanic cavity so that auditory ossicles vibrate freely. Osteopetrotic mice showed a smaller volume of the tympanic cavity but had larger auditory ossicles compared with controls. Most interestingly, the malleal processus brevis touched the medial wall of the tympanic cavity in osteopetrotic mice, which was also the case for c-Src kinase–deficient mice (with normal numbers of nonresorbing osteoclasts). 
Histological analysis and X-ray tomographic microscopy using synchrotron radiation showed that auditory ossicles in osteopetrotic mice were thicker and more cartilaginous than those in control mice. The auditory brainstem response showed that mice of both genotypes experienced hearing loss, and laser Doppler vibrometry revealed that the malleus behind the tympanic membrane failed to vibrate. We analyzed hearing function and morphological features of auditory ossicles in osteopetrotic mice, which lack osteoclasts because of the deficiency of either cytokine RANKL or transcription factor c-Fos. Little is known about whether and how bone-resorbing osteoclasts play a role in the vibration of auditory ossicles. In the middle ear, a chain of three tiny bones (ie, malleus, incus, and stapes) vibrates to transmit sound from the tympanic membrane to the inner ear. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
